Germany’s national laboratory for heavy-ion physics, GSI in Darmstadt, turns pure science to the service of humanity with a cancer treatment facility using carbon ions.
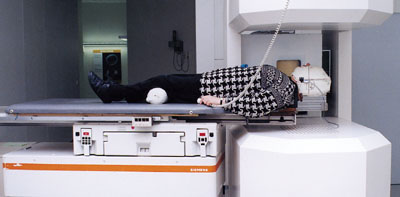
“A bridge has been established between fundamental research and its applications, ” said Hessen State Minister for Science and Art, Christine Hohmann-Dennhardt, speaking at the official inauguration of a new cancer therapy facility at the German national heavy-ion physics laboratory, GSI in Darmstadt, on 15 September. The facility uses carbon ions from GSI’s heavy-ion synchrotron, SIS, to target traditionally difficult-to-operate tumours. The first two patients were treated at GSI last December and are tumour-free. A further nine patients underwent a successful course of therapy in August.
Cancer is second only to cardiovascular disease in the grisly league table of fatal illness. Gene therapy holds out the hope for a cure in the long-term, but in the short-term traditional methods of surgery, chemotherapy and radiotherapy must be used. GSI’s new carbon-ion facility adds a new string to the radiotherapist’s bow, allowing tumours in sensitive areas to be targeted safely and effectively.
The idea of using particle beams for cancer therapy is not new. In 1946, Bob Wilson first realized the potential of the technique when he observed that, unlike photons or electrons, proton beams deposit most of their energy at the end of their paths in the so called Bragg peak. This opened up the possibility of targeting deep-seated tumours, or tumours close to sensitive organs, with much reduced risk to surrounding healthy tissue, by making the dose conform more closely to the volume of the tumour.
The first treatments were performed in 1954 when John Lawrence, Ernest Lawrence’s brother, treated patients with proton beams at Berkeley’s 184-inch synchrocyclotron. Three years later the same laboratory scored another first by turning helium ions to therapeutic use. And Berkeley’s pioneering role wasn’t confined to the United States. In 1956 Lawrence’s friend and colleague Cornelius Tobias was a guest scientist in Sweden. Working with Lars Leksel and Borje Larsson at Uppsala, he helped initiate a programme of surgery and therapy using protons from the University’s cyclotron.
Pioneering
Harvard’s cyclotron laboratory has the longest continuous history of using proton beams to treat patients. The first patients arrived in 1961 and the facility will remain in use until all treatments are transferred to the Northeast Proton Therapy Center, scheduled to receive its first patients in February 1999. Then, having treated over 7000 patients, the Harvard cyclotron will take a well-earned retirement.
The main limitation of these pioneering efforts has been the lack of techniques for shaping the beam Even today particle therapy units use mainly passive beam-shaping systems, such as absorbers or collimators, adapted from photon therapy. These shape the irradiated volume to match a target volume identified by tomographic imaging techniques. Passive methods, however, only permit the same limited conformation of dose to tumour that can be reached with conforming photon therapy.
The GSI facility is the first to provide extremely precise tumour conformity using magnetic beam scanning in two dimensions and by actively varying the energy of the accelerator to give the third dimension. Beam delivery is based on a novel, fully three dimensional, treatment planning system that takes into account the differences in biological efficiency of the beam in different tissues as well as so-called early and late effects. Early effects essentially tumour cell killing are maximized, while late effects complications in healthy tissue are minimized.
After Berkeley and Japan’s Heavy Ion Medical Accelerator, HIMAC, in Chiba, GSI becomes the world’s third ion therapy centre. The laboratory is a relative latecomer to the field, but development of the innovative beam delivery system and treatment planning based on a more profound understanding of radiobiological particle action took more than a decade of intense experimental work. When GSI switched on its first accelerator in 1975, radiation biology experiments were among the first to be performed. Their initial goal was to investigate the biological effects of cosmic radiation during space flight, but the results have been fed into the modern therapy programme. They showed, in what has come to be known as the “Darmstadt hook”, that the microscopic structure of the energy deposition process is far more important in determining biological effects than distinctions between biological systems.

GSI’s fully active beam-scanning system works on much the same principle as a television set, where the picture is built up of lines consisting of individual pixels. Instead of a flat, regular screen, the GSI beam plays on a three-dimensional, irregular tumour. The scanning process, however, is very similar. The tumour is mapped and divided into 3-D pixels, the number of necessary particles is calculated for each pixel, and the beam targeted at that pixel until the calculated number has been delivered.
The deepest layers are targeted first and the energy of the beam is then reduced by the accelerator to position the Bragg peak on successively shallower layers. This procedure requires extremely reliable accelerator operation guaranteeing spatial beam stability of better than one millimetre at the target. Energy and intensity are changed from pulse to pulse, which for the SIS means within a second. To guarantee the required accuracy, an independent control system monitors the beam’s position every 100 microseconds and its intensity every 10 microseconds. If either deviates by more than 2% from what is expected, the beam is shut off within half a millisecond.
Such strict operating conditions are far beyond what is required from the SIS in its regular research role, but routine patient treatment has shown that the accelerator is up to the task. Just a handful of interruptions per day have been provoked by the control system during treatments involving the targeting of tens of thousands of pixels.
GSI in profile
The Darmstadt GSI heavy-ion laboratory is most famous for its discoveries of transuranic elements numbers 107 to 112. These were filtered out from the fusion products resulting from collisions between a heavy-ion beam from the universal linear accelerator, UNILAC, and a target of lead or bismuth.
Element 107, named bohrium after Danish physicist Niels Bohr, was discovered in 1981. Hassium, named after the state of Hessen, followed in 1983, and meitnerium, named after Austrian physicist Lise Meitner, came in 1984. Elements 110 to 112 were discovered between 1994 and 1996 and have yet to be named.
Elements 113 and 114, a predicted hidden “valley of stability” are next on the laboratory’s hit-list. GSI’s main research theme is the investigation of hot, dense nuclear matter in the collisions of heavy ions with stationary targets.
This began in 1975 at the UNILAC which has today been joined by the SIS and the experimental storage ring, ESR. Heavy-ion research has implications for basic nuclear physics as well as for astrophysics and the properties of neutron stars. In
another strand of research using the ESR, scientists can strip off the electron shells from even very heavy atoms. This allows them to study quantum electrodynamics the most precise theory in physics to unprecedented levels of accuracy.
Although GSI is primarily a pure science laboratory, the tumour therapy work is not the only domain of applied physics under study there. Plasma physics, particularly with the goal of producing energy generation through heavy-ion-driven inertial confinement fusion, is also an important part of the laboratory’s work.





