Image credit: Ronald Fernando Garcia.
Magic numbers appear in nuclei in which protons or neutrons completely fill a shell. The existence of magic numbers to explain certain regularities observed in nuclei was discovered in 1949 independently by M Goeppert-Mayer and J D Jensen, who were awarded the Nobel prize in 1963. Nuclei containing a magic number of nucleons, namely 2, 8, 20, 28, 50 and 82, are spherical, and present a very high degree of stability, which makes them very difficult to excite. The degree of “magicity” of a nucleus can be determined by precisely determining its shape, mass, excitation energy and electromagnetic observables – properties that can be precisely studied with dedicated experiments at ISOLDE.
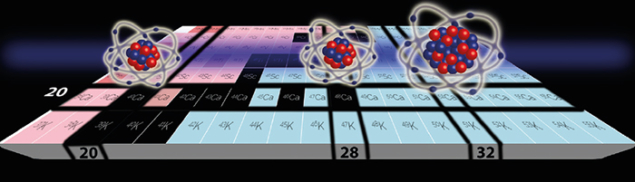
The calcium-isotopic chain (Z = 20, magic proton number) is a unique nuclear system to study how protons and neutrons interact inside of the atomic nucleus: two of its stable isotopes are magic in both their proton and neutron number (40Ca with N = 20 and 48Ca with N = 28). Despite an excess of eight neutrons, 48Ca exhibits the striking feature that it has an identical mean square charge radius as 40Ca. In addition, experimental evidence of doubly magic features in a short-lived calcium isotope, 52Ca (N = 32), was obtained in 2013 (Wienholtz et al. 2013 Nature 498 346). Therefore, to determine the radius beyond 48Ca was crucial from an experimental and theoretical point of view. The new determination of the nuclear radius is now challenging the magicity of the 52Ca isotope.

Image credit: ISOLDE/COLLAPS Collaboration.
The measurements were performed by using high-resolution bunched-beam collinear laser spectroscopy in the COLLAPS installation at ISOLDE, CERN. The charge radii for 40–52Ca isotopes were obtained from the measured optical isotope shifts extracted from the fit of the hyperfine experimental spectra. Indeed, although the average distance between the electrons and the nucleus in an atom is about 5000 times larger than the nuclear radius, the size of the nuclear-charge distribution is manifested as a perturbation of the atomic energy levels. A change in the nuclear size between two isotopes gives rise to a shift of the atomic hyperfine structure (hfs) levels. This shift between two isotopes, one million times smaller than the absolute transition frequency, commonly known as the isotope shift, includes a part that is proportional to the change in the nuclear mean square charge radii. Measurement of such a tiny change is only possible by using ultra-high-resolution techniques. With a production yield of only a few hundred ions per second, the measurement on 52Ca represents one of the highest sensitivities ever reached using fluorescence-detection techniques. The collinear laser spectroscopy technique developed at ISOLDE has been established as a unique method to reach such high resolution, and has been applied with different detection schemes to study a variety of nuclear chains.
The resulting charge radius of 52Ca is found to be much larger than expected for a doubly magic nucleus, and largely exceeds the theoretical predictions. The large and unexpected increase of the size of the neutron-rich calcium isotopes beyond N = 28 challenges the doubly magic nature of 52Ca, and opens new and intriguing questions on the evolution of nuclear size away from stability, which are of importance for our understanding of neutron-rich atomic nuclei.