The challenge of delivering cost-effective, high-precision treatment.
In 1946, accelerator pioneer Robert Wilson laid the foundation for hadron therapy with his article in Radiology about the therapeutic interest of protons for treating cancer (CERN Courier December 2006 p24). Sixty-five years later, proton therapy has grown into a mainstream clinical modality. More than 60,000 patients worldwide have been treated since the establishment of the first hospital-based treatment centre in Loma Linda, California, in 1990 and various companies are now offering turn-key solutions for medical centres. Moreover, encouraging studies with other types of hadrons have resulted in the creation and planning of various dedicated facilities.
Hadron therapy is the epitome of a multidisciplinary and transnational venture: its full development requires the competences of physicists, physicians, radiobiologists, engineers and IT experts, as well as collaboration between research and industrial partners. The translational aspects are extremely relevant because the communities involved are traditionally separate and they have to learn to speak the same “language”. Ions that are considered “light” by physicists, such as carbon, are “heavy” for radiobiologists – and this is just one of many examples.
Although state-of-the-art techniques borrowed from particle accelerators and detectors are increasingly being used in the medical field for the early diagnosis and treatment of tumours and other diseases, medical doctors and physicists lack occasions to get together and discuss global strategies. The first Physics for Health (PHE) workshop was organized in 2010 at CERN exactly to develop synergies between these diverse communities. Preparations are now underway for a follow-up workshop, which will join forces with the International Conference on Translational Research in Radiation Oncology (ICTR). The ICTR-PHE 2012 conference will be held in Geneva on 27 February – 2 March. The aim is to catalyse and enhance further exchanges and interactions between experts in this multidisciplinary field where medicine, biology and physics intersect.
The advantages of hadron therapy
The clinical interest in hadron therapy resides in the fact that it delivers precision treatment of tumours, exploiting the characteristic shape of the Bragg curve for hadrons, i.e. the dose deposition as a function of the depth of matter traversed. While X-rays lose energy slowly and mainly exponentially as they penetrate tissue, hadrons deposit almost all of their energy in a sharp peak – the Bragg peak – at the very end of their path.
The Bragg peak makes it possible to target a well defined cancerous region at a depth in the body that can be tuned by adjusting the energy of the incident particle beam, with reduced damage to the surrounding healthy tissues. The dose deposition is so sharp that new techniques had to be developed to treat the whole target. These fall under the categories of passive scattering, where one or more scatterers are used to spread the beam, and spot scanning, where a thin, pencil-like beam covers the target volume in 3D under the control of sweeping magnets coupled to energy variations.
While the advantages of protons over photons are quantitative in terms of the amount and distribution of the delivered dose, several studies show evidence that carbon ions damage cancer cells in a way that the cells cannot repair themselves. Carbon therapy may be the optimal choice to tackle radio-resistant tumours; other light ions, such as helium, are also being investigated.

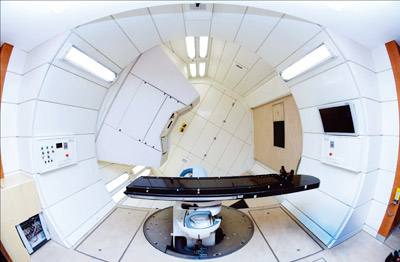
Image credit: CNAO.
Although hadron therapy has largely shown its potential scientifically, the relative complexity of the required infrastructures limits its exploitation. “Hadron therapy is not a replacement for conventional radiotherapy or surgery, but is an additional tool in the toolbox of the oncologists,” confirms Robert Miller of the Mayo Clinic in the US, which just embarked on the construction of two proton-therapy facilities. Indeed, hadron therapy is mostly used for treating tumours that are located close to vital organs that would be unacceptably damaged by X-rays, or in paediatric oncology, where quality of life and late side effects are a major concern.
At present, the world map of hadron therapy is divided into three distinct regions: Asia (mainly Japan), the US and Europe. In addition, three proton-therapy facilities are operational in Russia and one in South Africa.
Japan is the uncontested leader in treatment and clinical studies with carbon ions (CERN Courier June 2010 p22). By the end of 2010, its two major facilities – the Heavy-Ion Medical Accelerator in Chiba (HIMAC) and the Hyogo Ion Beam Medical Center – had treated more than 90% of the 6600 world total of patients irradiated with carbon ions. Clinical experience in the Japanese centres has not only demonstrated that carbon therapy is more effective than conventional photon radiotherapy on certain types of tumours but also that, with respect to both protons and photons, a significant reduction of the overall treatment time and the number of irradiation sessions can be achieved. In addition to the existing facilities, Japan is planning the construction of two more centres for carbon-ion therapy and two more for proton therapy. Following this lead, China and other countries in Asia have constructed or are planning several carbon-ion and proton-therapy facilities.
In the US alone more than 30,000 patients have already been treated with protons over the past 20 years, half of them at Loma Linda. There are currently six active proton facilities, three more under construction and a number of centres announced or planned in the near future. When hadron therapy was still confined to facilities operating within particle-physics laboratories, the US pioneered the use not only of protons but also of other ions: between 1957 and 1992, the Bevalac in Berkeley treated about 2500 cancer patients with particles including neon, carbon, silicon and argon. Today, there is no therapy centre delivering ions other than protons in America. Plans for the future include only an R&D facility in the San Francisco Bay area called SPARC, which would be a joint effort between Stanford/SLAC and Lawrence Berkeley National Laboratory/University of California San Francisco, and a carbon and helium facility at the Mayo Clinic.
Europe has 10 active proton facilities, with five more planned or under construction. Capitalizing on the experience gained from the carbon-therapy programme at GSI in Darmstadt and at Heidelberg, Europe is now witnessing the birth of “dual” centres that are capable of delivering beams of both protons and carbon ions. Two major centres were recently completed: Heidelberg Ion Therapy Centre (HIT), which started treatments at the end of 2009 and has irradiated about 500 patients with carbon ions to date; and the Centro Nazionale di Adroterapia Oncologica (CNAO) in Pavia, which started treating the first patient with protons in September and will launch the preclinical phase with carbon ions in the coming months. The MedAustron dual facility in Wiener Neustadt is currently under construction (CERN Courier October 2011 p33) and more centres of a similar nature are at different stages in planning and implementation in France and Germany.
HIT is the first facility in the world to be equipped with a gantry for carbon ions, i.e. a structure to rotate the particle beam and guide it to the patient at a chosen angle. Using the gantry, radio-oncologists can select the optimal beam direction to minimize the amount of healthy tissue traversed by the hadrons before reaching the tumour. They can also irradiate the target from multiple angles – a technique that, thanks to the overlapping beams, delivers to the target a total dose that is much higher than in the surrounding normal tissues. CNAO relies on an accelerator design implemented by Terapia con Radiazion Adroniche Foundation based on the results of the Proton Ion Medical Machine Study hosted at CERN from 1996 to 1999. The CNAO facility will deliver horizontal and vertical beams and a gantry will be added at a later stage.
Co-ordination and training
With the blossoming of carbon therapy in Europe, the European Network for Light Ion Therapy (ENLIGHT) considered that the time was right to leverage the experience at the various facilities, as well as the wealth of advances in beam delivery for conventional radiation therapy, and improve the technology with the aim of more effective and affordable cancer treatments with particles. While developing and optimizing the next-generation facilities remains the community’s primary goal, it is also of paramount importance that the existing centres collaborate intensively and that researchers, clinicians and patients have protocols to access these structures. Within this framework, the Union of Light Ion Centres in Europe (ULICE) project was launched in September 2009, funded by the European Commission.
ULICE is a collaboration of 20 partners led by Roberto Orecchia, scientific director of CNAO. The project involves all of the existing and planned European carbon-therapy facilities, including the two leading European companies in the hadron-therapy sector, IBA and Siemens. The participation of private companies ensures that specific issues related to possible future industrial production are addressed. IBA has designed and installed the majority of clinically operating proton-therapy facilities in the world and is developing innovative and more affordable single-room proton systems, as well as superconducting cyclotron solutions for carbon. Siemens Healthcare is one of the world’s largest providers of medical solutions and was the first company outside of Asia to enter the carbon-ion therapy market. The company delivered the complete patient environment at HIT and the treatment-planning system at CNAO.
ULICE is a four-year project built around three pillars: Joint Research Activities that focus on development of instruments and protocols; Networking, to increase co-operation between facilities and research communities wanting to work with the research infrastructure; and Transnational Access, which aims at allowing researchers to use the facilities and for radiobiological and physics experiments to take place.
At the recent mid-term review meeting in Marburg, Richard Pötter, a radiation oncologist at the University of Vienna and co-ordinator of the Joint Research Activities of ULICE, confirmed that the first achievements of the research work are extremely encouraging. The existing clinical-study protocols worldwide have been reviewed to start defining common guidelines for patient selection. Specific studies have focused on setting up appropriate structures for a comprehensive and prospective multicentre clinical-research programme and the development of a dosimetry protocol. Important steps forward have also been made in defining uniform methods and concepts for irradiation doses and tumour volumes in radio-oncology, to create a common language not only within the consortium but across all of the communities involved in different forms of radiotherapy. The ULICE consortium is working hard to develop new concepts for more compact and affordable gantries: the HIT gantry is a steel giant of 25 m in length and 13 m in height, and alternative designs are clearly needed.
Within the activities of Transnational Access, the ULICE partners are examining the complex task of setting up a structure to allow access to the existing European facilities for patients, clinical and experimental research, as well as for clinical training and education. Japan is once again an example to follow, with the International Open Laboratory (IOL) programme of the National Institute of Radiological Sciences launched in 2008 to grant beam time at HIMAC to external researchers. There are currently four active IOLs with Columbia University, Colorado State University, the University of Sussex, Karolinska Institutet and GSI. As of summer 2011, researchers from eligible countries can apply to take part in research activities or submit experimental proposals in the clinical, radiobiological and physical field at the University Hospital of Heidelberg and at CNAO. In the words of Jürgen Debus, medical director of the Department of Radiation Oncology and Radiation Therapy of Heidelberg University Hospital and co-ordinator of the ULICE Transnational Access: “A technology has worth in the medical field only if it is spread and if everyone can participate to its evolution with their experience and feedback.” Applications for participation in the Transnational Access programme will be reviewed by a multicentre scientific committee and successful applicants will be granted free access thanks to the European Union Transnational Access funding. In the same framework, ULICE is also developing an international web-based documentation and data-management system, which will be an essential tool for transnational and multicentre clinical studies in particle therapy.
In the coming years, the project will focus on expanding and consolidating the transnational access and on developing innovative gantry designs. The support of ENLIGHT will be instrumental to dissemination, communication and networking, which will help it reach out to the widest possible community.

Image credit: Nathalie Hospital for ENLIGHT.
ENLIGHT also actively supports the creation of the next generation of the necessary highly specialized experts through the Particle Training Network for European Radiotherapy (PARTNER), funded by the European Commission under the Marie Curie Initial Training Network programme (CERN Courier March 2010 p27). Both ENLIGHT and PARTNER are co-ordinated by Manjit Dosanjh at CERN. PARTNER is offering research and training opportunities in leading European institutions and companies to 25 young researchers who are mostly involved in PhD studies at the same time. At the recent annual meeting in Marburg, the presentations of the individual projects displayed clearly the variety of topics being addressed and the quality of the research. PARTNER is now in its fourth and final year, and in a few months it will be time to review the results that have been achieved.
• For more about ULICE, see http://cern.ch/ULICE.






