Peter Thirolf, Benedict Seiferle and Lars von der Wense describe how recent progress in understanding thorium’s nuclear structure, and new upcoming results, could enable an ultra-accurate nuclear clock with applications in fundamental physics.

For the past 60 years, the second has been defined in terms of atomic transitions between two hyperfine states of caesium-133. Such transitions, which correspond to radiation in the microwave regime, enable state-of-the art atomic clocks to keep time at the level of one second in more than 300 million years. A newer breed of optical clocks developed since the 2000s exploit frequencies that are about 105 times higher. While still under development, optical clocks based on aluminium ions are already reaching accuracies of about one second in 33 billion years, corresponding to a relative systematic frequency uncertainty below 1 × 10–18.
To further reduce these uncertainties, in 2003 Ekkehard Peik and Christian Tamm of Physikalisch-Technische Bundesanstalt in Germany proposed the use of a nuclear instead of atomic transition for time measurements. Due to the small nuclear moments (corresponding to the vastly different dimensions of atoms and nuclei), and thus the very weak coupling to perturbing electromagnetic fields, a “nuclear clock” is less vulnerable to external perturbations. In addition to enabling a more accurate timepiece, this offers the potential for nuclear clocks to be used as quantum sensors to test fundamental physics.
Clockwork
A clock typically consists of an oscillator and a frequency-counting device. In a nuclear clock (see “Nuclear clock schematic” figure), the oscillator is provided by the frequency of a transition between two nuclear states (in contrast to a transition between two states in the electronic shell in the case of an atomic clock). For the frequency-counting device, a narrow-band laser resonantly excites the nuclear-clock transition, while the corresponding oscillations of the laser light are counted using a frequency comb. This device (the invention of which was recognised by the 2005 Nobel Prize in Physics) is a laser source whose spectrum consists of a series of discrete, equally spaced frequency lines. After a certain number of oscillations, given by the frequency of the nuclear transition, one second has elapsed.

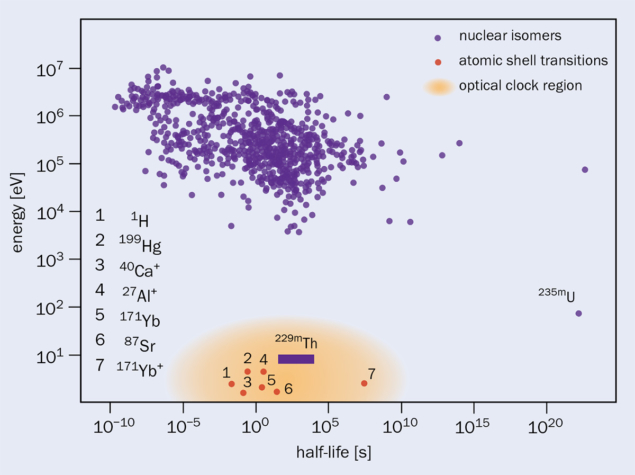
The need for direct laser excitation strongly constrains applicable nuclear-clock transitions: their energy has to be low enough to be accessible with existing laser technology, while simultaneously exhibiting a narrow linewidth. As the linewidth is determined by the lifetime of the excited nuclear state, the latter has to be long enough to allow for highly stable clock operation. So far, only the metastable (isomeric) first excited state of 229Th, denoted 229mTh, qualifies as a candidate for a nuclear clock, due to its exceptionally low excitation energy.
The existence of the isomeric state was conjectured in 1976 from gamma-ray spectroscopy of 229Th, and its excitation energy has only recently been determined to be 8.19 ± 0.12 eV (corresponding to a vacuum-ultraviolet wavelength of 151.4 ± 2.2 nm). Not only is it the lowest nuclear excitation among the roughly 184,000 excited states of the 3300 or so known nuclides, its expected lifetime is of the order of 1000 s, resulting in an extremely narrow relative linewidth (ΔE/E ~ 10–20) for its ground-state transition (see “Unique transition” figure). Besides high resilience against external perturbations, this represents another attractive property for a thorium nuclear clock.
Networks of ultra-precise synchronised nuclear clocks could enable a search for ultra light dark matter
Achieving optical control of the nuclear transition via a direct laser excitation would open a broad range of applications. A nuclear clock’s sensitivity to the gravitational redshift, which causes a clock’s relative frequency to change depending on its absolute height, could enable more accurate global positioning systems and high-sensitivity detections of fluctuations of Earth’s gravitational potential induced by seismic or tectonic activities. Furthermore, while the few-eV thorium transition emerges from a fortunate near-degeneracy of the two lowest nuclear-energy levels in 229Th, the Coulomb and strong-force contributions to these energies differ at the MeV level. This makes the nuclear-level structure of 229Th uniquely sensitive to variations of fundamental constants and ultralight dark matter. Many theories predict variations of the fine structure constant, for example, but on tiny yearly rates. The high sensitivity provided by the thorium isomer could allow such variations to be identified. Moreover, networks of ultra-precise synchronised clocks could enable a search for (ultra light) dark-matter signals.
Two different approaches have been proposed to realise a nuclear clock: one based on trapped ions and another using doped solid-state crystals. The first approach starts from individually trapped Th ions, which promises an unprecedented suppression of systematic clock-frequency shift and leads to an expected relative clock accuracy of about 1 × 10–19. The other approach relies on embedding 229Th atoms in a vacuum–ultraviolet (VUV) transparent crystal such as CaF2. This has the advantage of a large concentration (> 1015/cm3) of Th nuclei in the crystal, leading to a considerably higher signal-to-noise ratio and thus a greater clock stability.
Precise characterisation
A precise characterisation of the thorium isomer’s properties is a prerequisite for any kind of nuclear clock. In 2016 the present authors and colleagues made the
first direct identification of 229mTh by detecting electrons emitted from its dominant decay mode: internal-conversion (IC), whereby a nuclear excited state decays by the direct emission of one of its atomic electrons (see “Isomeric signal” figure). This brought the long-term objective of a nuclear clock into the focus of international research.
Currently, experimental access to 229mTh is possible only via radioactive decays of heavier isotopes or by X-ray pumping from higher-lying rotational nuclear levels, as shown by Takahiko Masuda and co-workers in 2019. The former, based on the alpha decay of 233U (2% branching ratio), is the most commonly used approach. Very recently, however, a promising new experiment exploiting β– decay from 229Ac was performed at CERN’s ISOLDE facility led by a team at KU Leuven. Here, 229Ac is online-produced and mass-separated before being implanted into a large-bandgap VUV-transparent crystal. In both population schemes, either photons or conversion electrons emitted during the isomeric decay are detected.
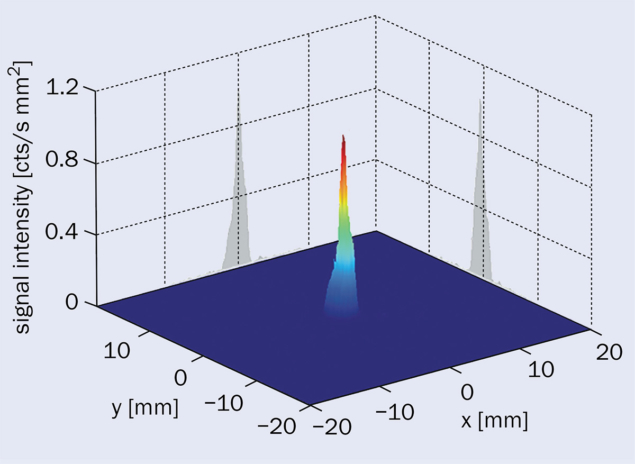
In the IC-based approach, a positively charged 229mTh ion beam is generated from alpha-decay daughter products recoiling off a 233U source placed inside a buffer-gas stopping cell. The decay products are thermalised, guided by electrical fields towards an exit nozzle, extracted into a longitudinally 15-fold segmented radiofrequency quadrupole (RFQ) that acts as an ion guide, phase-space cooler and optionally a beam buncher, followed by a quadrupole mass separator for beam purification. In charged thorium isomers, the otherwise dominant IC decay branch is energetically forbidden, leading to a prolongation of the lifetime by up to nine orders of magnitude.
Operating the segmented RFQ as a linear Paul trap to generate sharp ion pulses enables the half-life of the thorium isomer to be determined. In work performed by the present authors in 2017, pulsed ions from the RFQ were collected and neutralised on a metal surface, triggering their IC decay. Since the long ionic lifetime was inaccessible due to the limited ion-storage time imposed by the trap’s vacuum conditions, the drastically reduced lifetime of neutral isomers was targeted. Time-resolved detection of the low-energy conversion electrons determined the lifetime to be 7 ± 1 μs.
Excitation energy
Recently, considerable progress has been made in determining the 229mTh excitation energy – a milestone en route to a nuclear clock. In general, experimental approaches to determine the excitation energy fall into three categories: indirect measurements via gamma-ray spectroscopy of energetically low-lying rotational transitions in 229Th; direct spectroscopy of fluorescence photons emitted in radiative decays; and via electrons emitted in the IC decay of neutral 229mTh. The first approach led to the conjecture of the isomer’s existence and finally, in 2007, to the long-accepted value of 7.6 ± 0.5 eV. The second approach tries to measure the energy of photons emitted directly in the ground-state decay of the thorium isomer.
The first direct measurement of the thorium isomer’s excitation energy was reported by the present authors and co-workers in 2019. Using a compact magnetic-bottle spectrometer equipped with a repulsive electrostatic potential, followed by a microchannel-plate detector, the kinetic energy of the IC electrons emitted after an in-flight neutralisation of Th ions emitted from a 233U source could be determined. The experiment provided a value for the excitation energy of the nuclear-clock transition of 8.28 ± 0.17 eV. At around the same time in Japan, Masuda and co-workers used synchrotron radiation to achieve the first population of the isomer via resonant X-ray pumping into the second excited nuclear state of 229Th at 29.19 keV, which decays predominantly into 229mTh. By combining their measurement with earlier published gamma-spectroscopic data, the team could constrain the isomeric excitation energy to the range 2.5–8.9 eV. More recently, led by teams at Heidelberg and Vienna, the excited isomers were implanted into the absorber of a custom-built cryogenic magnetic micro-calorimeter and the isomeric energy was measured by detecting the temperature-induced change of the magnetisation using SQUIDs. This produced a value of 8.10 ± 0.17 eV for the clock-transition energy, resulting in a world-average of 8.19 ± 0.12 eV.
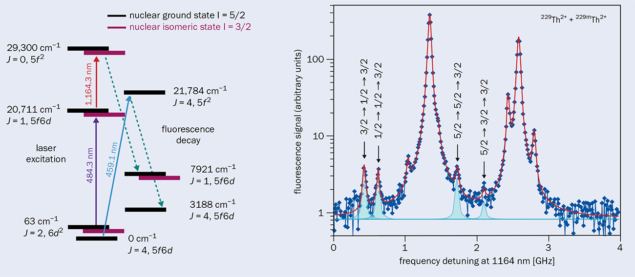
Besides precise knowledge of the excitation energy, another prerequisite for a nuclear clock is the possibility to monitor the nuclear excitation on short timescales. Peik and Tamm proposed a method to do this in 2003 based on the “double resonance” principle, which requires knowledge of the hyperfine structure of the thorium isomer. Therefore, in 2018, two different laser beams were collinearly superimposed on the 229Th ion beam, initiating a two-step excitation in the atomic shell of 229Th. By varying both laser frequencies, resonant excitations of hyperfine components both of the 229Th ground state and the 229mTh isomer could be identified and thus the hyperfine splitting signature of both states could be established by detecting their de-excitation (see “Hyperfine splitting” figure). The eventual observation of the 229mTh hyperfine structure in 2018 not only will in the future allow a non-destructive verification of the nuclear excitation, but enabled the isomer’s magnetic dipole and electrical quadrupole moments, and the mean-square charge radius, to be determined.
Roadmap towards a nuclear clock
So far, the identification and characterisation of the thorium isomer has largely been driven by nuclear physics, where techniques such as gamma spectroscopy, conversion-electron spectroscopy and radioactive decays offer a description in units of electron volts. Now the challenge is to refine our knowledge of the isomeric excitation energy with laser-spectroscopic precision to enable optical control of the nuclear-clock transition. This requires bridging a gap of about 12 orders of magnitude in the precision of the 229mTh excitation energy, from around 0.1 eV to the sub-kHz regime. In a first step, existing broad-band laser technology can be used to localise the nuclear resonance with an accuracy of about 1 GHz. In a second step, using VUV frequency-comb spectroscopy presently under development, it is envisaged to improve the accuracy into the (sub-)kHz range.
Another practical challenge when designing a high-precision ion-trap-based nuclear clock is the generation of thermally decoupled, ultra-cold 229Th ions via laser cooling. 229Th3+ is particularly suited due to its electronic level structure, with only one valence electron. Due to the high chemical reactivity of thorium, a cryogenic Paul trap is the ideal environment for laser cooling, since almost all residual gas atoms will freeze out at 4 K, increasing the trapping time into the region of a few hours. This will form the basis for direct laser excitation of 229mTh and will also enable a measurement of the not yet experimentally determined isomeric lifetime of 229Th ions. For the alternative development of a compact solid-state nuclear clock it will be necessary to suppress the 229mTh decay via internal conversion in a large band-gap, VUV transparent crystal and to detect the γ decay of the excited nuclear state. Proof-of-principle studies of this approach are currently ongoing at ISOLDE.
Laser-spectroscopy activities on the thorium isomer are also ongoing in the US, for example at JILA, NIST and UCLA
Many of the recent breakthroughs in understanding the 229Th clock transition emerged from the European Union project “nuClock”, which terminated in 2019. A subsequent project, ThoriumNuclearClock (ThNC), aims to demonstrate at least one nuclear clock by 2026. Laser-spectroscopy activities on the thorium isomer are also ongoing in the US, for example at JILA, NIST and UCLA.
In view of the large progress in recent years and ongoing worldwide efforts both experimentally and theoretically, the road is paved towards the first nuclear clock. It will complement highly precise optical atomic clocks, while in some areas, in the long run, nuclear clocks might even have the potential to replace them. Moreover, and beyond its superb timekeeping capabilities, a nuclear clock is a unique type of quantum sensor allowing for fundamental physics tests, from the variation of fundamental constants to searches for dark matter.
Further reading
A Ludlow et al. 2015 Rev. Mod. Phys. 87 637.
T Masuda et al. 2019 Nature 573 238.
E Peik et al. 2021 Quantum Sci. Technol. 6 034002.
E Peik and C Tamm 2003 Eur. Phys. Lett. 61 181.
B Seiferle et al. 2019 Nature 573 243.
B Seiferle et al. 2017 Phys. Rev. Lett. 118 042501.
T Sikorsky et al. 2020 Phys. Rev. Lett. 125 142503.
J Thielking et al. 2018 Nature 556 321.